Post-Streptococcal Glomerulonephritis – Causes, Symptoms and Treatment
Highlights of Post-Streptococcal Glomerulonephritis
- Post-streptococcal glomerulonephritis(PSGN) is an inflammation of the kidneys that occurs after throat or skin infection, affecting primarily children aged 5-12.
- Malnutrition and weakened immune systems increase the risk of PSGN. It can be more common in lower socioeconomic areas and certain ethnic groups.
- Swelling that begins around the eyes and increased blood pressure are the most common symptom of this condition.
- In severe cases of kidney damage, kidney replacement therapy with hemodialysis or peritoneal dialysis may be needed in the treatment of PSG
- The outlook of post-streptococcal glomerulonephritis is generally good, especially for children, who usually have complete recovery.
What is Post-Streptococcal Glomerulonephritis?
Post-streptococcal glomerulonephritis (PSGN) is a kidney disorder that usually occurs as a throat or skin infection complication with specific strains of Group A streptococcus bacteria. This condition is a form of glomerulonephritis, where the tiny filtering units of the kidney, called glomeruli, become inflamed and damaged, leading to impaired kidney function.
It is often seen in children one to two weeks after a throat infection or up to six weeks after a skin infection. [1] It is often seen worldwide but is more prevalent in developing countries and areas with limited access to healthcare, where streptococcal infections are more widespread.
Causes and Risk Factors of PSGN
The causes and factors predisposing to PSGN are:

- Immune reaction: After an infection, the body’s immune system creates antibodies to fight off the strep infection. These antibodies are meant to target the bacteria, but because parts of the bacteria look similar to molecules in the kidney, the antibodies also end up attacking the kidney. This attack on the kidney leads to inflammation and damage, which causes the symptoms of PSGN. [2]
- Infections: PSGN can also result from other infections, including bacterial (endocarditis, enterocolitis, pneumonia), viral (hepatitis B and C, HIV, CMV, Epstein Barr virus, parvovirus B19), fungal (coccidioidomycosis, histoplasmosis), and parasitic (malaria, leishmania, toxoplasmosis, schistosomiasis) sources. [3]
- Age: PSGN primarily affects children aged 5-12 years, is rare in adults, but can affect the elderly. [4]
- Immunity: PSGN can occur in older adults with weakened immune systems due to conditions like diabetes or cancer. Malnutrition can weaken the immune system, making children more prone to infections that trigger PSGN. [5]
- Socioeconomic Status: PSGN is more prevalent in communities with lower socioeconomic status. Children from these backgrounds tend to experience higher rates of the disease. [6]
- Genetic Factors: A family history of PSGN increases your risk of developing the condition. [5]
- Geography and Ethnicity: PSGN is of significant concern in developing countries and rural areas, like northwestern Ontario, where Indigenous and rural communities are particularly affected. In places like New Zealand and Australia, children of Pacific Island, Māori, and Torres Strait Islander descent have higher rates compared to European or other populations.[7,8]
Symptoms of PSGN
Around 50% of patients with PSGN have no symptoms. Common symptoms include:
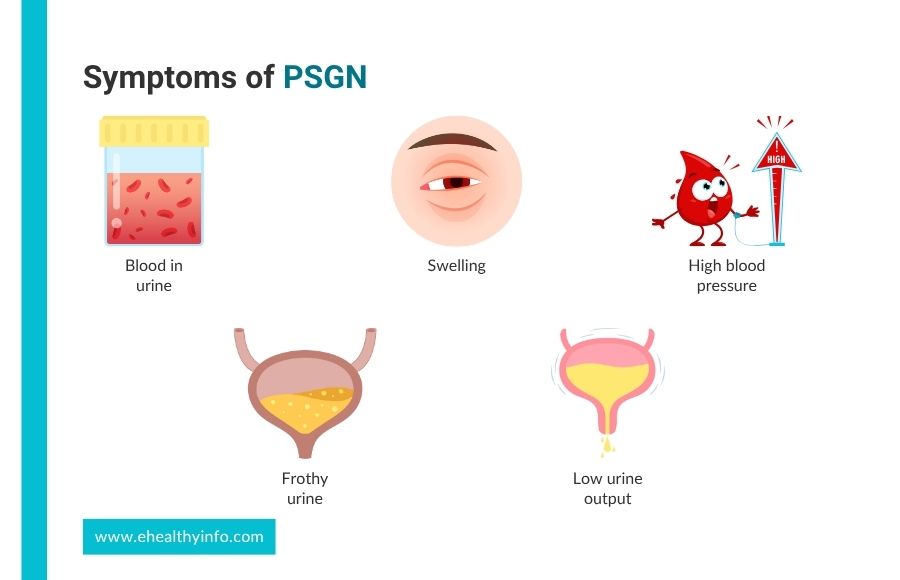
1. Blood in urine
This is the most common symptom of PSGN. The inflammation and damage can cause red blood cells to leak into your urine. This could cause your urine to appear tea-colored, cola-colored, smoky, or rusty. [9] Blood may not always be visible to the naked eye but can be detected through lab tests.
2. Swelling
Swelling from PSGN typically starts one to two weeks after infection. It usually begins around the eyes, may spread to other body parts like ankles or belly, and is most noticeable in the morning but decreases by evening. In severe cases, fluid may build up in the lungs, leading to breathing problems. [3]
3. High blood pressure
In PSGN, when your kidneys cannot filter blood properly, your body can retain sodium and water, which may increase blood pressure and sometimes cause headaches. [10]
4. Frothy urine
In PSGN, damage to the kidney allows the protein to leak through, making the urine appear frothy and bubbly. [11]
5. Low urine output
When kidney function drops, urine output may fall below 400 mL daily in adults. In severe cases, it can be less than 100 mL per day.
Diagnosis of PSGN
The diagnoses of PSGN are:
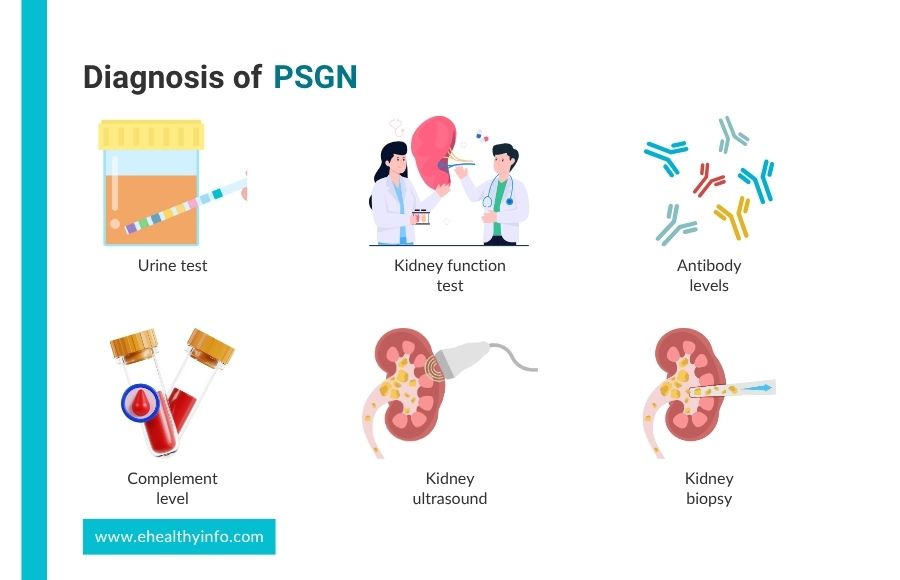
1. Urine test
It is essential to the diagnosis and helps detect red blood cells and protein in your urine. A dipstick test can detect protein in the urine, and the levels are usually moderate but significantly higher in PSGN. [12]
2. Blood tests
- Kidney function test: This test is used to find the functioning of the kidney, which is done by measuring creatinine and blood urea nitrogen. Elevated levels of both can indicate reduced kidney function due to PSGN. [13]
- Antibody levels: Antistreptolysin O is an antibody the immune system produces in response to an infection. A high ASO titer indicates a recent strep infection. Other antibody tests, like Anti-DNase B, support the diagnosis of a recent infection if the ASO titer is not elevated. [14]
- Complement level: Complement proteins like C3 are part of the immune system and help fight infections. In PSGN, immune complexes deposit in the kidneys, leading to inflammation. This process consumes C3, often leading to low levels in the blood. [15]
3. Kidney ultrasound
This imaging test may be performed to evaluate the size and structure of the kidney and rule out other possible causes of kidney dysfunction. In PSGN, the kidneys may appear slightly enlarged due to inflammation. [11]
4. Kidney biopsy
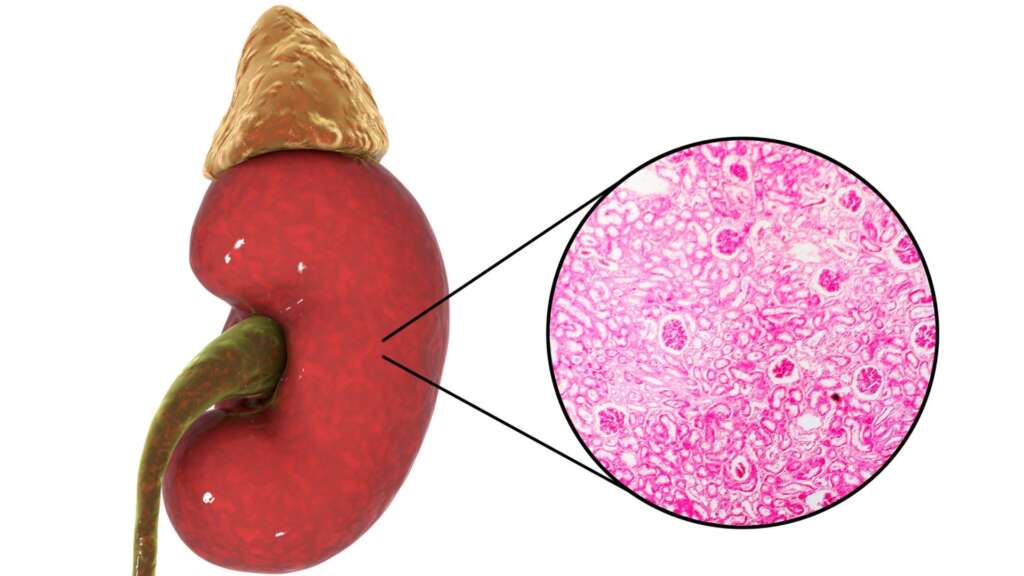
A kidney biopsy is not routinely performed in PSGN. However, a biopsy might be done if the diagnosis is uncertain or if the patient has an atypical presentation, like severe symptoms in adults with rapidly worsening conditions. This involves taking a small piece of kidney tissue that would typically show the deposited immune complexes in the glomeruli, inflammation, and damage that is happening there. [3,10,16]
Treatment of PSGN
PSGN treatment focuses on eradicating streptococcal infection, managing symptoms, and preventing complications.
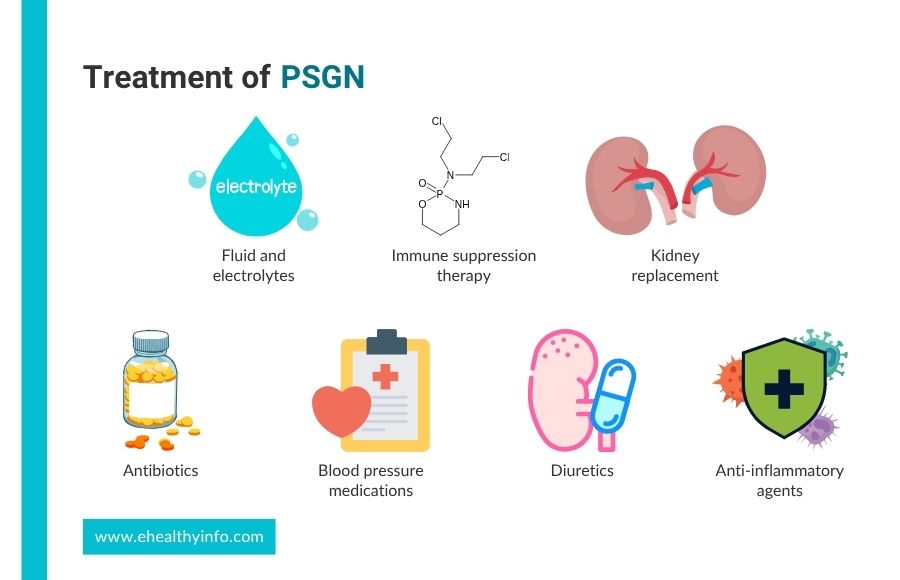
1. Medications
- Antibiotics: If there is an ongoing streptococcal infection at diagnosis, antibiotics like penicillin or amoxicillin are given to clear the infection and prevent its spread. However, antibiotics do not reverse the kidney damage already caused by PSGN. In some instances, prophylactic antibiotics may be considered to prevent recurrent streptococcal infections, particularly in high-risk populations. [16]
- Blood Pressure Medications: Medications like captopril, enalapril, and valsartan may control blood pressure by relaxing blood vessels and reducing the pressure within glomeruli. [17]
- Diuretics: Medications like furosemide help your body get rid of excess fluid, reduce swelling, and lower blood pressure.[16]
- Anti-inflammatory agents: In most cases of PSGN, anti-inflammatory medications like corticosteroids are not used, as the inflammation in the kidneys typically resolves on its own. [16]However, your doctor might consider their use in rare, severe cases with persistent kidney inflammation.[16]
2. Fluids and electrolytes
Due to swelling, limiting fluid intake may be necessary and should be restricted to match the insensible loss and urine output. Reducing salt intake can help you manage fluid retention and high blood pressure, which are common issues in PSGN. [18]
3. Immune Suppression Therapy
In rare cases of severe, rapidly worsening kidney damage from PSGN, treatments like cyclophosphamide might be used. These medicines help calm the immune system but are used carefully and only if needed. [19]
4. Kidney Replacement
If severe acute kidney failure doesn’t improve with standard treatments, kidney replacement therapy like hemodialysis or peritoneal dialysis might be needed. This therapy helps manage fluid buildup and electrolyte imbalances when kidney function is very poor.[16]
Complications of PSGN
PSGN can cause various complications, both short-term and long-term. The most common include:

- Kidney injury: PSGN can lead to mild kidney issues in some children, known as AKI, and in severe cases, it may lead to long-term kidney damage, needing a transplant. [16]
- Blood Pressure: High blood pressure is a common complication and often needs treatment with diuretics or other medications. [20]
- Effect on heart and lungs: Severe high blood pressure and fluid overload can cause congestive heart failure and pulmonary edema. In rare cases, OSGN can cause diffuse alveolar hemorrhage, leading to respiratory distress requiring aggressive management. [21]
- Chronic Kidney Disease: PSGN has a good prognosis, but a small proportion of people may develop chronic kidney disease, with persistent proteinuria, hypertension, and impaired function of the kidneys. [21]
PSGN and Recovery of Kidneys
The outlook for post-streptococcal glomerulonephritis is usually good, especially in children who often recover fully with normal kidney function. [22]In the long term, most people do well and don’t develop chronic kidney disease. [21] In rare cases, PSGN can cause severe kidney injury and require ongoing treatment and monitoring. [22] Overall, PSGN is usually not life-threatening, and most people recover well. [22]
PSGN and Malnutrition
Malnutrition can worsen PSGN and affect its progression. Low levels of albumin in the blood, often from malnutrition, can further reduce kidney function in PSGN patients. Studies show that 37.3% of children with PSGN had low albumin levels, which is linked to worse kidney function. [13] Malnutrition weakens the immune system, making people more likely to get infections, including the streptococcal infections that cause PSGN. It can delay recovery and increase the risk of complications like persistent protein in the urine and high blood pressure.[20]
Questions To Ask Your Doctor
- How long will it take for my kidneys to recover fully?
- How often will I need to test my kidney health?
- Will I need to take medications long-term?
- How would I know my condition is getting worse?
- Does PSGN recur in the future?
References
- Rawla P, Padala SA, Ludhwani D. Poststreptococcal Glomerulonephritis. Prim Care Update Ob Gyns 2022;10:24–8. https://doi.org/10.1016/S1068-607X(02)00141-5.
- Alhamoud MA, Salloot IZ, Mohiuddin SS, AlHarbi TM, Batouq F, Alfrayyan NY, et al. A Comprehensive Review Study on Glomerulonephritis Associated With Post-streptococcal Infection. Cureus 2021;13:e20212–e20212. https://doi.org/10.7759/cureus.20212.
- Iyengar A, Kamath N, Radhakrishnan J, Estebanez BT. Infection-Related Glomerulonephritis in Children and Adults. Semin Nephrol 2023;43:151469. https://doi.org/10.1016/j.semnephrol.2023.151469.
- Herthelius M, Berg U. Renal function during and after childhood acute poststreptococcal glomerulonephritis. Pediatric Nephrology 1999;13:907–11. https://doi.org/10.1007/s004670050725.
- Kambham N. Postinfectious Glomerulonephritis. Adv Anat Pathol 2012;19:338–47. https://doi.org/10.1097/PAP.0b013e31826663d9.
- Vogel AM, Lennon DR, van der Werf B, Diack M, Neutze JM, Horsfall M, et al. Post‐streptococcal glomerulonephritis: Some reduction in a disease of disparities. J Paediatr Child Health 2018;55:652–8. https://doi.org/10.1111/jpc.14263.
- Wong W, Morris MC, Zwi J. Outcome of severe acute post-streptococcal glomerulonephritis in New Zealand children. Pediatric Nephrology 2009;24:1021–6. https://doi.org/10.1007/s00467-008-1086-5.
- Chong HC, Hung T, Hohls A, Francis JR, Chaturvedi S. Clinical characteristics of hospitalised children with acute post‐streptococcal glomerulonephritis in the Top End of Australia. J Paediatr Child Health 2023;59:735–42. https://doi.org/10.1111/jpc.16386.
- Pais PJ, Kump T, Greenbaum LA. Delay in Diagnosis in Poststreptococcal Glomerulonephritis. J Pediatr 2008;153:560–4. https://doi.org/10.1016/j.jpeds.2008.04.021.
- Brant Pinheiro SV, de Freitas VB, de Castro GV, Rufino Madeiro BC, de Araújo SA, Silva Ribeiro TF, et al. Acute Post-Streptococcal Glomerulonephritis in Children: A Comprehensive Review. Curr Med Chem 2022;29:5543–59. https://doi.org/10.2174/0929867329666220613103316.
- Bajracharya P, Khadgi A, Shrestha S, Silwal R, Tandukar A. Acute Post-streptococcal Glomerulonephritis in a Pediatric Population: A Five-Year Retrospective Study. Cureus 2024. https://doi.org/10.7759/cureus.56082.
- Hamadah AM, Gharaibeh K, Mara KC, Thompson KA, Lieske JC, Said S, et al. Urinalysis for the diagnosis of glomerulonephritis: role of dysmorphic red blood cells. Nephrology Dialysis Transplantation 2018;33:1397–403. https://doi.org/10.1093/ndt/gfx274.
- Demircioglu Kılıc B, Akbalık Kara M, Buyukcelik M, Balat A. Pediatric post-streptococcal glomerulonephritis: Clinical and laboratory data. Pediatr Int 2018;60:645–50. https://doi.org/10.1111/PED.13587.
- Maness DL, Martin M, Mitchell G. Poststreptococcal Illness: Recognition and Management. Am Fam Physician 2018;97:517–22.
- Abugrain K, McCulloch MI, Muloiwa R, Luyckx VA, Buys H. A 6-year review of acute post-streptococcal glomerulonephritis at a public children’s hospital in Cape Town, South Africa. Pediatr Nephrol 2024;39:1809–16. https://doi.org/10.1007/s00467-023-06247-8.
- Beck LH, Ayoub I, Caster D, Choi MJ, Cobb J, Geetha D, et al. KDOQI US Commentary on the 2021 KDIGO Clinical Practice Guideline for the Management of Glomerular Diseases. American Journal of Kidney Diseases 2023;82:121–75. https://doi.org/10.1053/j.ajkd.2023.02.003.
- VanDeVoorde RG. Acute Poststreptococcal Glomerulonephritis: The Most Common Acute Glomerulonephritis. Pediatr Rev 2015;36:3–13. https://doi.org/10.1542/pir.36.1.3.
- Duong MD, Reidy KJ. Acute Postinfectious Glomerulonephritis. Pediatr Clin North Am 2022;69:1051–78. https://doi.org/10.1016/j.pcl.2022.08.001.
- Kanai H, Sawanobori E, Koizumi K, Ohashi R, Higashida K. Pediatric case of crescentic post-streptococcal glomerulonephritis with myeloperoxidase anti-neutrophil cytoplasmic antibody. Clin Nephrol 2014. https://doi.org/10.5414/cn108286.
- Ayoob RM, Schwaderer AL. Acute Kidney Injury and Atypical Features during Pediatric Poststreptococcal Glomerulonephritis. Int J Nephrol 2016;2016:1–5. https://doi.org/10.1155/2016/5163065.
- Dhakal AK, Shrestha D, Singh SK, Acharya S. Clinical profile of children with acute post-streptococcal glomerulonephritis. Pediatric Nephrology 2023;38:3327–36. https://doi.org/10.1007/s00467-023-05982-2.
- Skrzypczyk P, Ofiara A, Zacharzewska A, Pańczyk-Tomaszewska M. Acute post-streptococcal glomerulonephritis – immune-mediated acute kidney injury – case report and literature review. Central European Journal of Immunology 2021;46:516–23. https://doi.org/10.5114/ceji.2021.112244.
Return to Home